
Thermal Overman Rearrangement of a Glucal Derivative
A case study by Franz Amann - Senior Scientist Development at CARBOGEN AMCIS

Driven by the economy of scale, the size of production vessels should be increased with the targetedproduct amount. As a consequence, scale dependent requirements such as mixing and heat transfer within the vessel become more challenging. It is important to control both, processing times and reaction conditions. However, syntheses on large scale may face technical limits and may also be less controllable with regards to release of heat in a strongly exothermic reaction. If such challenges already arise at laboratory scale experiments, a scale-up will be difficult if not impossible.
This is where the flow chemistry approach can have a significant impact on how organic syntheses are executed. Flow reactors on a meso or micro scale allow to handle challenges related to temperature, pressure or concentration and will help to optimize the production process. As an additional advantage, safety risks of a synthesis can be reduced as starting materials react only in small amounts at any time. Flow chemistry complements conventional production processes by widening the possible range of parameters (process intensification) and opens the doors for new chemical prospects. On the other hand, its specific features may not fit for all reaction types e.g. very slow conversions or highly heterogenic systems. Therefore each project needs to be assessed individually in order to identify the right strategy.
A new approach to improve production processes at CARBOGEN AMCIS
At CARBOGEN AMCIS, flow chemistry becomes more and more relevant when finding new ways to expand large scale production capabilities. In a recent customer project with Actelion Pharmaceuticals Ltd., a thermal Overman Rearrangement of a glucal derivative, performed in a tube reactor on pilot plant scale, was one of the key steps. This example supports not only the flow chemistry approach, but also shows on how the different CARBOGEN AMCIS sites can work together and benefit from specific site characteristics.
All process development activities for flow chemistry on laboratory scale (> 100 g) were performed at the CARBOGEN AMCIS site in Hunzenschwil, Switzerland. CARBOGEN AMCIS in Manchester (UK) has designed an appropriate production reactor and manufactured at large scale once the process has been successfully developed and transferred.
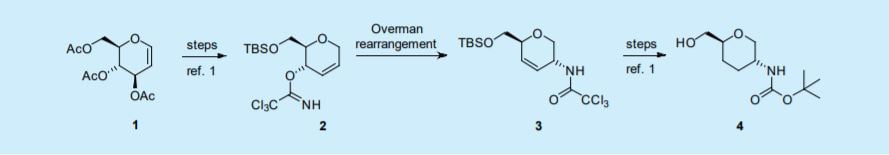
In that case, commercially available tri-O-acetyl-D-glucal (1) is converted in four steps to the
O-[ (2R, 3S)-2-(tertbutyldimethylsilyloxymethyl)-3, 6-dihydro-2H-pyran-3-yl]-trichloro-ethanecarboximidate (2) and then into the trichloroacetamide (3) by a thermal Overman rearrangement. This reaction is key to obtain Actelion’s intermediate (4).
The trichloroacetamide could be isolated in good yield and purity on small scale from a flask process.
The variable yields upon moderate scale-up and safety concerns with the exothermic decomposition of starting material and product, required a redesign of the process to enable multi kilogram production of the trichloroacetamide intermediate. As batch mode was not an option for scale-up, the development of a continuous flow process for the rearrangement was therefore considered the best option for process safety and suitability to produce the required quantities of the intermediate.
From small laboratory scale to a robust flow chemistry at large scale
The flow chemistry process has initially been developed with an HPLC pump and an electrically heated Hastelloy coil with a volume of 2.2 ml. Laboratory scale experiments demonstrated that the flow chemistry process could avoid the degradation of the trichloroacetamide. The new reaction conditions were within the DSC-decomposition zone for both, the starting material and the product but only the short residence time of ca. 2 min has prevented losses. The process conditions also allowed operation above the boiling point of the solvent. Thus, the toluene solution from the previous step could be directly processed for the flow reaction and used as is in the next conversion. The flow chemistry step could be seamlessly integrated into the existing synthesis.
In the second stage, a large scale flow chemistry process has been developed. The larger set-up consisted of a metering pump and two stainless steel coils with a volume of 250 mL (heating in a silicon oil bath) resp. 125 mL (rapid cooling). The reaction trials at kilo-lab scale outlined that the optimum running conditions at 220 °C and 12 bar in toluene were achieved with a flow rate of 80-85 mL/min or conversion of 20 g starting material in toluene per minute. A total of 95.3 kg was converted in ca. 84 hours of operation. Almost complete conversion could be achieved at a residence time of 2.8 min, which is only slightly higher than in the lab runs. Ultimately, crude trichloroacetamide was obtained in quantitative yield and with acceptable purity for downstream processing.
Successful site transfer and a fully developed continuous process
To conclude, an efficient continuous process for the high temperature Overman Rearrangement to obtain a trichloroacetamide has been developed. This process was demonstrated on multi kilogram scale with high yields (>90%). The subsequent transfer from small scale laboratory equipment to production at another site required only minor technical adjustments of the reaction parameters due to wider tubing and a different approach to keep the system pressure. The flow mode successfully mitigated the risks associated with the thermolabile substrate/product and is suitable for manufacturing scale. Additionally, it allowed using a convenient solvent for a high temperature reaction.
