
Tech Transfer Pharma: How to Successfully move from Clinical to Commercial Manufacturing?
The transition from clinical to commercial manufacturing is an important milestone in any pharma project. A successful tech transfer pharma strategy is critical in ensuring that your processes scale efficiently, are compliant, and maintain product quality. But how do you achieve this? Many companies underestimate the complexity of drug product tech transfer. This often leads to delays, increased costs, and inefficiencies in the process.
This article is also featured in the European Biopharmaceutical Review (Spring issue). You can read the original publication here. In this article, we discuss the principles of successful tech transfer and how you can achieve it quickly.
What is Tech Transfer in Pharma?
Technology transfer is often considered to be just an information transfer. However, it is much more than that. Tech transfer is an information transfer that is critical in ensuring that all the knowledge gained during product development is successfully transferred into commercial manufacturing.
What are the major challenges in Tech Transfer Services?
The major challenges in tech transfer services include:
- Aligning Development Data with Commercial Requirements
- Managing Time-to-Market Pressures without Increasing Risk
- Ensuring Access to Representative Materials
- Avoiding Gaps Between Sending and Receiving Sites
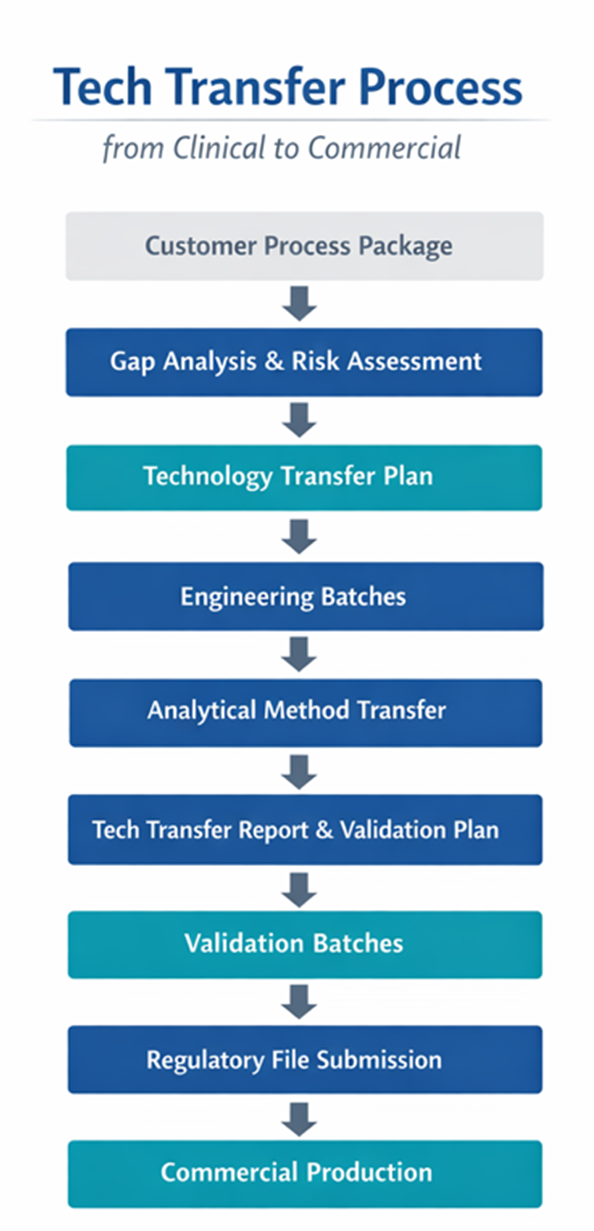
The major steps in Drug Product Tech Transfer
For a successful tech transfer pharma project, you need to follow these key steps:
- Gap Analysis & Risk Assessment
- Transfer Planning
- Execution & Validation
- Documentation and Regulatory Readiness
Each step is critical to a smooth and successful transfer process that meets regulatory requirements.
How to Accelerate Tech Transfer Without Increasing Risk?
Time is of the essence, yet rushing into tech transfer can cause more harm than good in the long run.
How can drug product tech transfer be accelerated without increasing risk? Companies can achieve this in the following ways:
- Utilize standard equipment and existing platforms
- Plan early and align expectations
- Work closely together
The key to successful and efficient execution is planning and preparation, not cutting corners or rushing through the process.
Scaling Up: Where Do Companies Often Experience Issues?
As drug products go from clinical to commercial scale, they pose a number of technical challenges.
In processes such as lyophilisation, scaling is not linear and equipment can have a big impact on results. That is why process understanding is key to a successful tech transfer for pharma companies.
Why Integration is Key to a Successful Tech Transfer for Pharma Companies?
When working with a tech transfer services provider, being integrated is key to a successful drug product tech transfer process.
When development, transfer, and manufacture are aligned, this results in:
- Reduced risks
- Faster project completion
- More robust processes
When tech transfer is treated as a strategic activity and not a final step, this makes all the difference to a successful tech transfer process.
-
Want the Full Step-By-Step Framework?
In this article, we have covered the basics of drug product tech transfer and pharma tech transfer services. However, for a successful tech transfer process, it is important to have a more in-depth understanding of:
Risk-based strategies
GMP and Annex 1
Real-life scale-up challenges
Best practice for ‘right first time’ execution
Authors
Julie Pagenaud, Pharmaceutical Operations Director, CARBOGEN AMCIS
Elea Ney, Technical Transfer Manager, CARBOGEN AMCIS
